Are botox injections safe 100%
Botox (Botulinum toxin), also known as botulinum endotoxin, is a bacterial endotoxin secreted by the deadly botulinum toxin and is highly toxic. Botulinum toxin acts on the endings of cholinergic motor nerves and antagonizes calcium ions in some way The role of botulinum toxin interferes with the release of acetylcholine from the motor nerve endings, so that muscle fibers cannot contract and cause muscle relaxation to achieve the purpose of wrinkle removal and beauty.
Botox is a neurotoxin, botulinum toxin prevents the nerve from entering the muscle being injected. You no longer need to contract that muscle.

Are botox injections safe?
Botulinum toxin is considered safe from 1989-2003 the FDA reported.
• 1437 adverse events
• 217 serious consequences
• 28 dead
You should know botulinum toxin has the following effects:
1.Wrinkle removal: general crow’s feet, forehead lines, brow lines , Nasal lines and neck wrinkles can be removed.
2. Slimming: The principle of using botulinum toxin injection for masseter muscle face-lifting is different from that of improving facial wrinkles.
3. Slim legs.
4. Raise the upper lip and alar muscles.
5. Improve unsightly eyebrows.
Certain parts of the body have very low levels of botulinum toxin. You need to make sure that the person injecting the medicine is experienced and competent.
Botulinum toxin type A (BTA) (Botox) was approved by the U.S. Food and Drug Administration (FDA) in 1989 for the treatment of strabismus and blepharospasm, in 2000 for the treatment of cervical dystonia, and in 2002 Get a cosmetic treatment for wrinkles (Botox Cosmetic). In 2002 alone, approximately 1.1 to 1.6 million people used cosmetic BTAs. Our goal was to review adverse events (AEs) reported to the FDA following BTA administration.
Reviewed all (therapeutic and cosmetic use) serious (under FDA regulations) AEs (from December 1989 to May 2003) reported to the FDA since the product was licensed (December 1989 to May 2003) and Nonserious AEs reported from December 2001 to November 2002. Report to FDA through the MedWatch system.
A total of 1437 adverse event reports were reviewed; 406 (217 severe and 189 severe) after therapeutic use of BTA and 1031 (36 severe and 995 severe) cosmetic use. The reported adverse events occurred mainly in female patients, with a median age of 50 years. Between December 2001 and November 2002, when severe and non-severe reports were assessed together, the proportion of reports classified as severe was 33 times higher for therapeutic reports than for cosmetic reports. The 217 serious AEs reported in treated cases involved a wide range of events, including all 28 reported deaths. Among cosmetic users, no deaths were reported, and of the 36 serious AEs, 30 possible complications were included in the FDA-approved label. The remaining 6 serious AEs did not show a pattern suggesting a common causal relationship to BTA. Among the 995 reported cosmetic cases, non-serious adverse events were the most common, including ineffectiveness (623, 63%), injection site reactions (190, 19%) and ptosis (111, 11%).
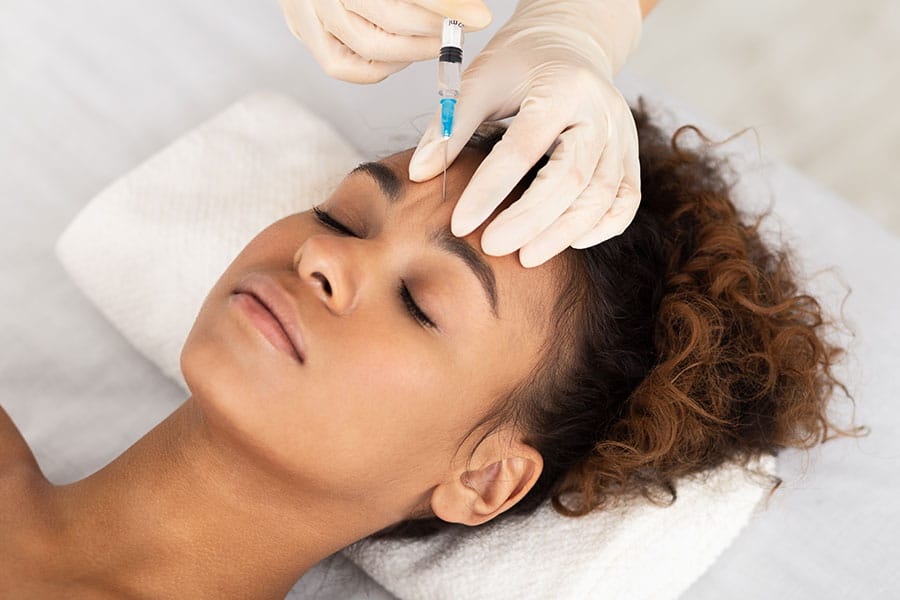
Serious adverse events were reported for therapeutic rather than cosmetic use, which may be related to higher doses, complicated underlying disease, or both. Few serious AEs were reported in cosmetic cases, and these were primarily events previously recognized for marker use in clinical trials of BTA. This study was primarily limited by the incompleteness of adverse event reporting by clinicians. These AE reports indicate that there are many differences from FDA-approved recommendations for drug dosage, dilution, handling, injection site, and storage.
So,Botulinum toxin injection is relatively safe, but you must choose to go to a regular beauty salon for treatment. Also, don’t worry about it right after the treatment. It will be natural after recovery. If it has already recovered, it may need to be repaired.